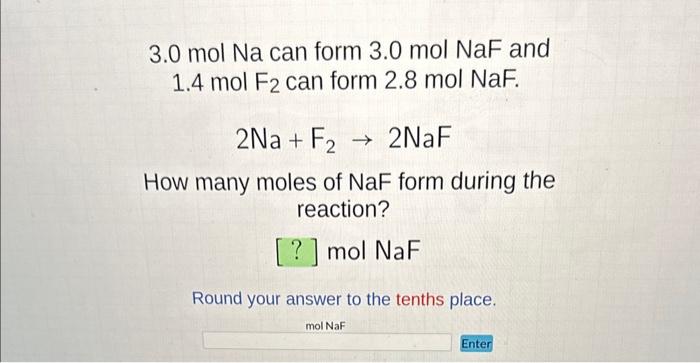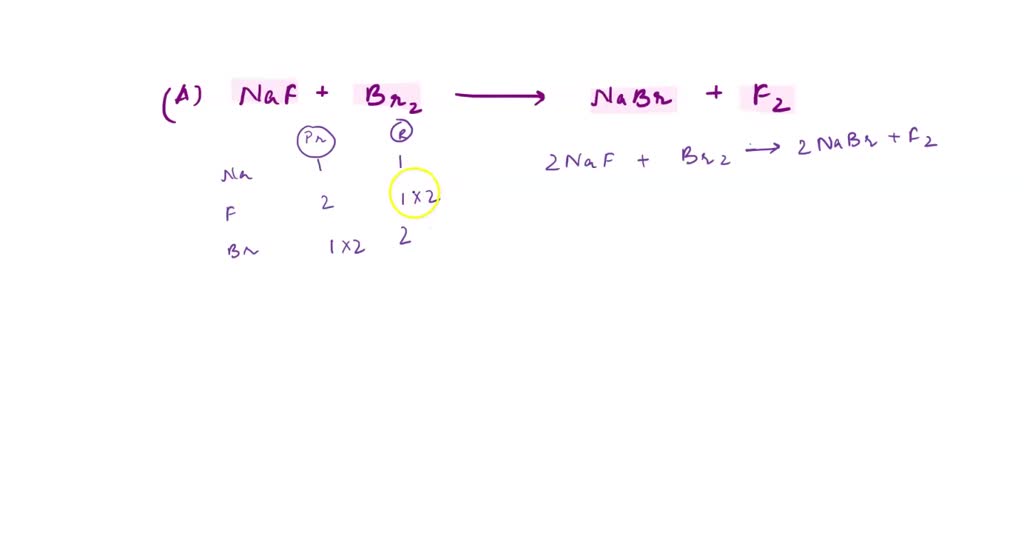
SOLVED: ' NaF + Br2 NaBr + F2 right answers only NaF NaBr Na= F= Na= F= Use equation to choose the correct coefficients from the answers below: 2,1,2,1 b. 3,2,1,1 1,2,2,1
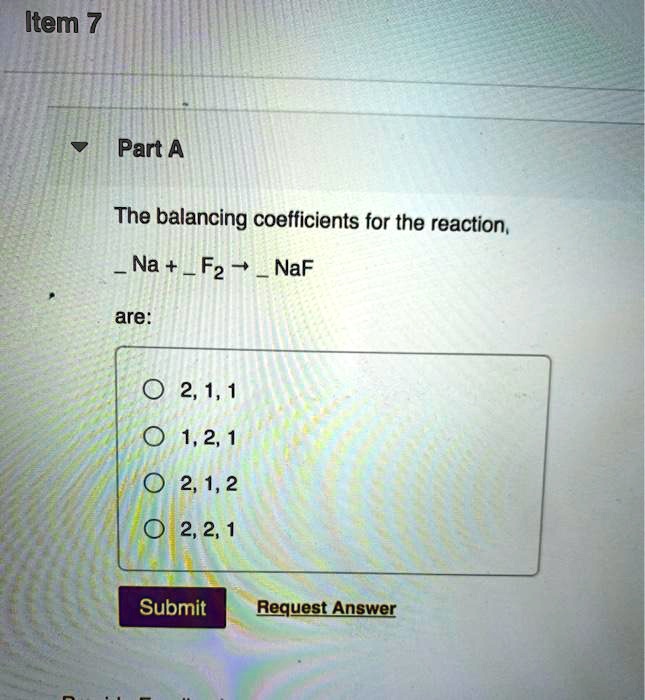
SOLVED: Item 7 Part A The balancing coefficients for the reaction, Na + F2 NaF are: 2,1,1 0 1,2,1 2,1,2 2,2,1 Submit Request Answer

Formation of NaF‐Rich Solid Electrolyte Interphase on Na Anode through Additive‐Induced Anion‐Enriched Structure of Na+ Solvation - Wang - 2022 - Angewandte Chemie International Edition - Wiley Online Library

How do you calculate the lattice energy of NaF using the Born-Haber cycle? (Delta Hsub for sodium is 107.5 kJ/mol) | Homework.Study.com
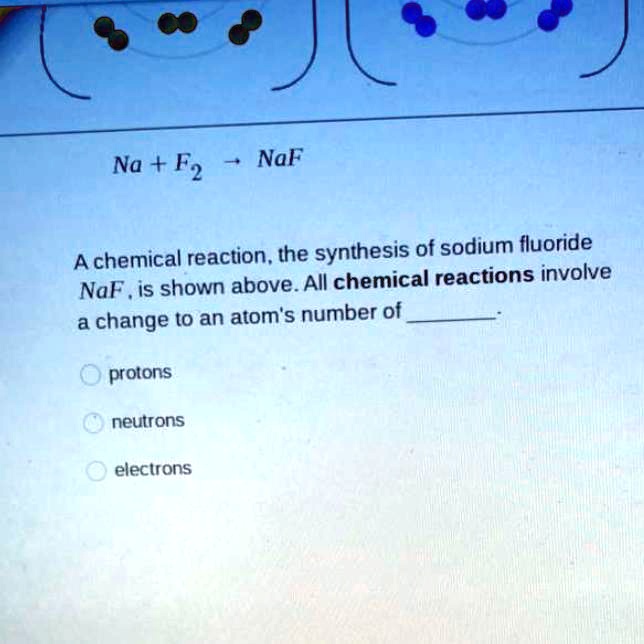
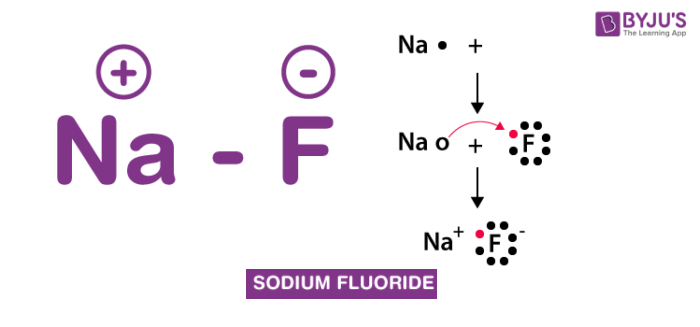
SOLVED: "na+F2-> naF a chemical reaction. the synthesis of sodium fluoride noF is shown above all chemical reactions involve a change to an atoms number of protons neutrons electrons NaF Na +

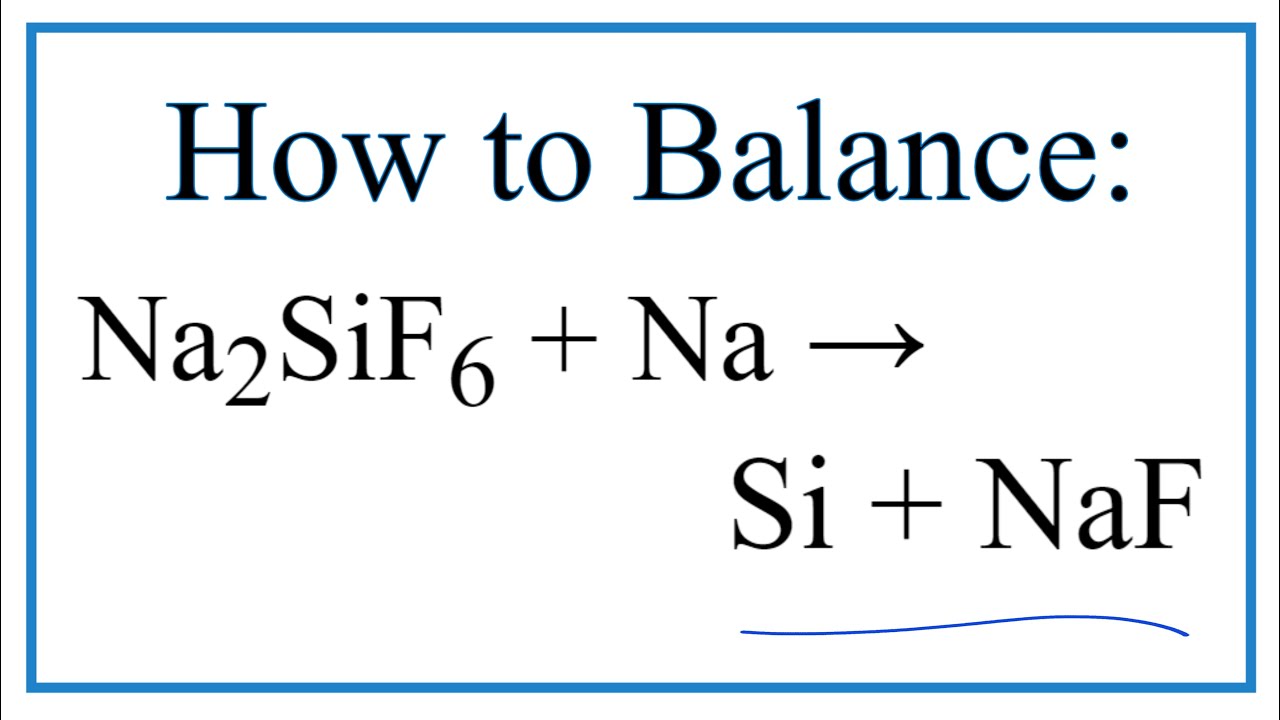




/cloudfront-us-east-1.images.arcpublishing.com/gray/EPJN2Q4OP5GP7P24DEYNKP2U5U.jpg)